Blog
BioClinica and ERT to Merge: Perspectives on Potential Synergies

On December 10, 2020, ERT, a clinical end-point data solutions company, announced its merger with BioClinica, a clinical trial management and imaging solutions company. The goal of the resulting enterprise will be to integrate the best of both worlds – ERT’s expertise in electronic Clinical Outcomes Assessment (eCOA), therapeutic expertise in cardiac safety and respiratory, and clinical endpoint measurement through wearables, with BioClinica’s expertise in imaging and clinical trial management solutions. The merger will equip the combined company to deliver data analytics, insights, business intelligence, virtual patient visits, and technology solutions to its clients.
In analyzing this development, we’ve taken a look at the hottest topics in the life sciences industry right now – decentralized and virtual clinical trials.
Virtual clinical trials – a revolution catalyzed by the pandemic
A virtual clinical trial is one in which certain parts of the clinical trial are conducted outside a clinical site, such as patient consent capture, trial data capture, or patient monitoring through sensors or wearables. The benefits to the pharmaceutical company include cost savings, better patient recruitment and retention, and improved data quality.
Earlier this year, we published a blog predicting that the 2020s would be the decade of virtual trials. It seems we were way off the mark – by about nine years. The year 2020 has already seen its fair share of virtual trials, as clinical trials that were put on pause due to lockdown restrictions were rescued by being converted to fully virtual or hybrid trials, such as cases in which clinical experts visited patients at their residences to collect vitals or samples, reducing delays.
#NoGoingBack
The virtual trial momentum isn’t temporary, and there’s increasing focus on virtual trials, even among investors. Not only this, many in the industry have pledged to preserve the progress they’ve made in clinical research due to the pandemic, including virtual trials.
On the same day the news of the merger was announced, the Decentralized Trials & Research Alliance (DTRA) was formed to unite stakeholders with a mission to make clinical trial participation widely accessible by advancing policies, research practices, and new technologies in decentralized patient-focused clinical research. Companies that are part of this alliance include technology vendors such as Medidata Solutions and Oracle Health Sciences, pharma companies such as Pfizer and Roche, CROs such as Parexel and Syneos Health, and others such as Amazon and the US Food and Drug Administration.
M&A and investment activity has increased, too. For example, Medable and Science 37 each received funding during the pandemic to advance their virtual trial offerings. And in November 2020, VirTrial, a telehealth platform for managing decentralized and virtual clinical trials, was acquired by Signant Health, a Clinical Trial Management System (CTMS) vendor, thus augmenting its virtual trial capabilities.
Clearly, virtual trials are a ripe area for M&A and investment activity given their disruptive capabilities and benefits. And we continue to expect more acquisitions, funding, and collaboration in this space in the near future.
What this all means for the merger
Our recently concluded PEAK Matrix assessment on clinical development platforms pointed out that BioClinica’s Cloud platform for clinical development does not have the capability to support virtual trials; we said it needed to invest in remote monitoring and eCOA capabilities to deliver on virtual trials. However, the solution does have a broad set of capabilities in the clinical, regulatory, and safety value chains.
As a result of the merger, however, BioClinica will be able to offer virtual trial capabilities to clients. ERT is one of the leading eCOA providers and through its wearable and sensor data capture capabilities, it is well positioned to conduct virtual trials in certain therapy areas. And it will be able to use the BioClinica Cloud offering to give clients a holistic clinical development experience, a win-win-win for ERT, BioClinica, and their clients.
Exhibit 1 shows the combined solution landscape.
Exhibit 1: The merger synergies
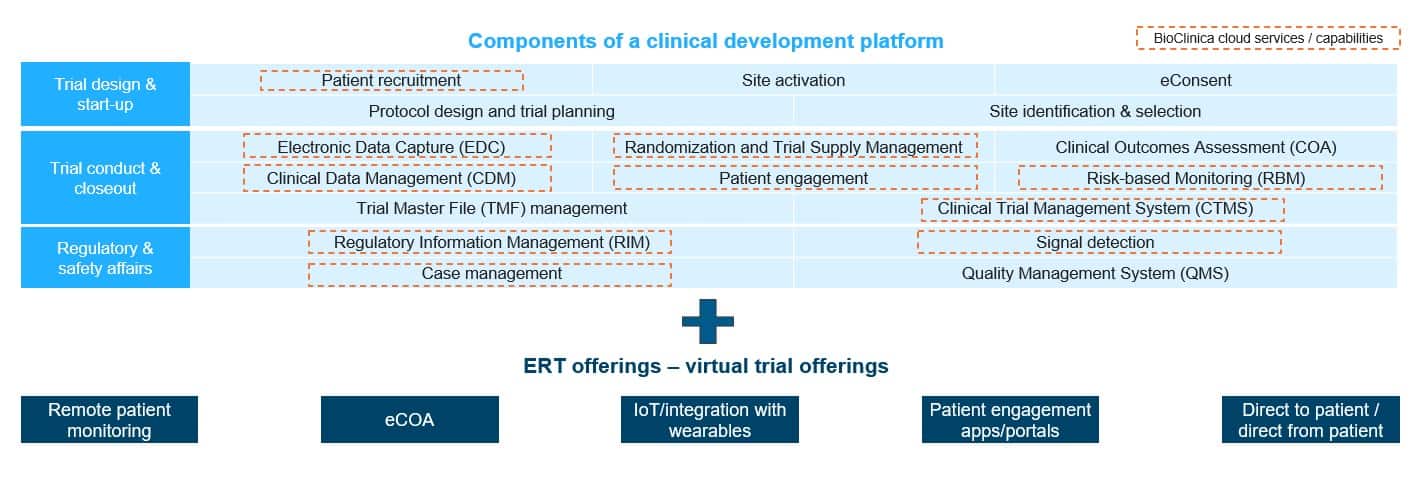
The merged entity will be able to showcase an end-to-end clinical development platform with enabling layers for virtual trial conduct. This move is definitely the right direction, at the most opportune time, and is just another sign of increasing interest in decentralized and virtual trials.
What are your views on this merger? Let us know your thoughts at [email protected] and [email protected].